Who We Are
Our Vision
Drastically change the face of surgical oncology so that every patient goes home cancer free
"My wife died of breast cancer in 2003. Since then, I have made it my mission to help prevent others from experiencing the same pain from cancer left behind after surgery."
W. David Lee, Co-Founder
Our Mission
At Lumicell, our mission is to improve the way cancer surgery is performed, starting with breast cancer. Our innovative, fluorescence-guided imaging technology is designed to illuminate cancerous tissue inside the body cavity following initial lumpectomy to help surgeons achieve a more complete surgical resection.
Goal of Surgical Resection
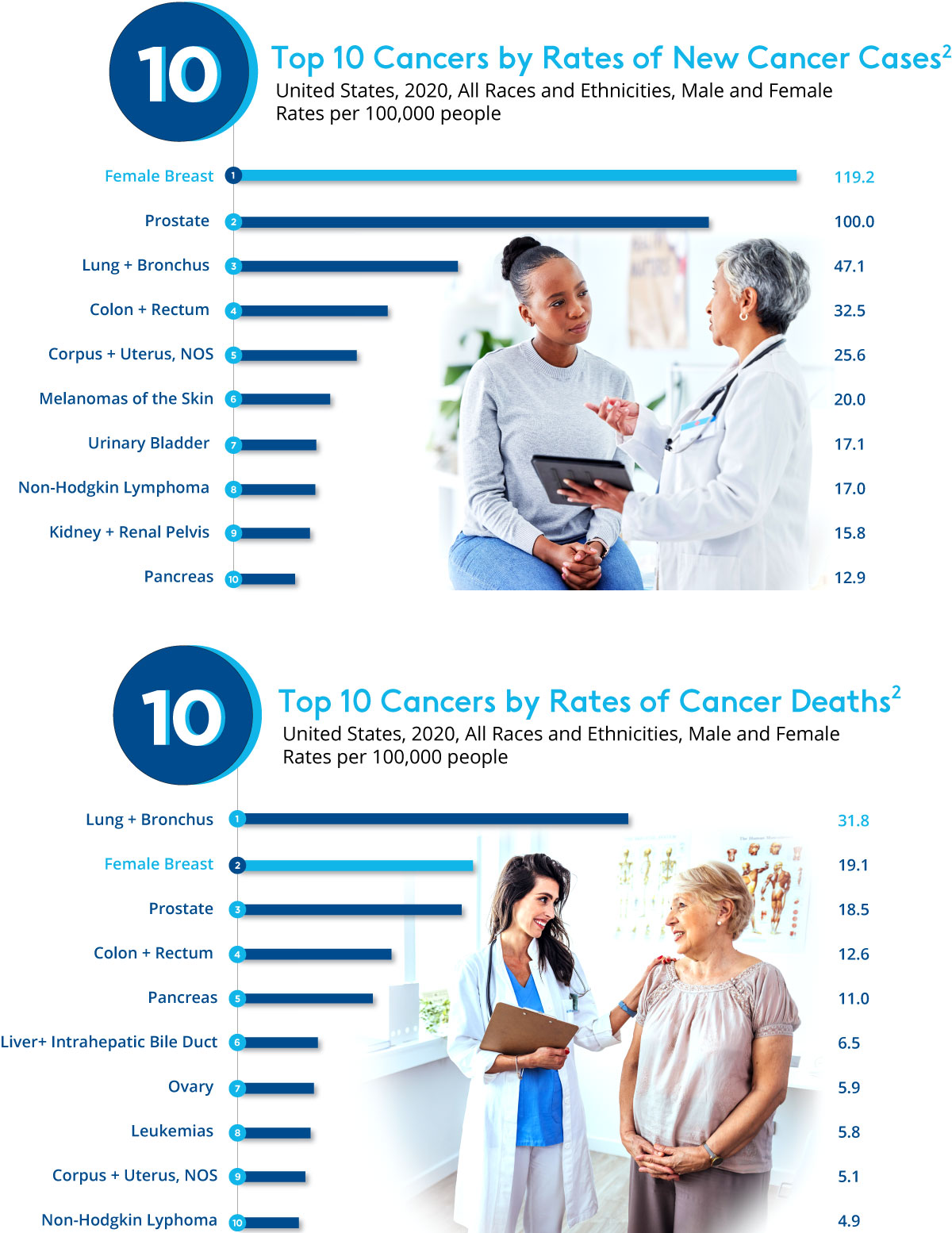
Cancer incidence in the US is increasing and is expected to cross 2 Million patients in 2024. There are multiple treatments for cancers with surgery as a predominant treatment. The goal of surgical resection is the eradication of cancer-both gross and microscopic.1
We believe that more can and should be done for surgical oncology care.
Imagine if surgeons could see and remove cancer cells in real-time during surgery
Lumicell recognizes the challenges surgeons face in finding and removing cancerous tissue while conserving as much healthy tissue as possible. That’s why we are committed to pioneering new technologies to help surgeons make more informed decisions in real-time.
Leadership Team

Jorge Ferrer, PhD
As a co-inventor of Lumicell’s drug/device technology, Jorge played a pivotal role in translating the innovations from his work in the lab of MIT’s Professor Moungi Bawendi (2023 Nobel Laurate in Chemistry) to founding Lumicell. He has led Lumicell from early-stage R&D through clinical development, overseeing the company’s PMA and NDA submissions and culminating in the successful FDA approval of its flagship breast cancer product. As Chief Scientific Officer, Jorge leads the strategic direction of technology development, spearheads the expansion into new cancer indications, and drives exploration of applications beyond oncology. He holds a doctorate in biological engineering and a M.S. in mechanical engineering from the Massachusetts Institute of Technology, and a B.S. in mechanical engineering from the University of Puerto Rico Mayagüez.

Elisabeth Carroll
Elisabeth Carroll is strategic global human resources professional with 20 years experience aligning employee performance with business strategy. She has an ability to quickly build strong partnerships with diverse leadership styles in a broad range of industries with demonstrated capability to work strategically and operationally in parallel. Previously, Elisabeth served as VP of Human Resources at Sensei Biotherapeutics to build the HR function and scale the organization after going public. At Quanterix she oversaw Human Resources as the organization scaled quickly, went public and acquired and integrated two organizations. She has deep experience supporting R&D as well as Commercialized global organizations within biotech start ups through large pharma. Elisabeth holds a Bachelors degree in Art History from Simmons University.

Daniel K. Harris, PhD

Brian Schlossberg, PhD
Brian joined Lumicell in 2018 as Director of Medical Affairs and is currently responsible for leading development of new indications for Lumicell’s fluorescence imaging technologies. He brings over 15 years of product development experience from medical device start-ups. Prior to joining Lumicell, Brian served as Director of Research and Development at Osteonovus; a medical device company developing biomaterials for spine and orthopedic indications. Brian earned his PhD in Biomedical Engineering from The University of New South Wales, where he studied in the Surgical & Orthopaedic Research Laboratories under Professor Bill Walsh.

Kate Smith
Board of Directors
Carlos Gonzalez
Founder, Rivas Capital
Andrey Zarur, PhD
Executive Chairman,
Lumicell
CEO, GreenLight Biosciences
David Furneaux
CEO, BlueIO & Managing Partner,
Kodiak Venture Partners
Elon Boms
Launch Capital, LLC
Kevin Krenitsky, MD
Former COO & CCO,
Foundation Medicine
Jim Alecxih
CEO, Virtual Incision
Le positionnement adopté par twincasino mise sur l'équilibre entre divertissement et sécurité. Les évolutions techniques régulières témoignent d'un investissement continu dans la qualité. Une foire aux questions très complète couvre la majorité des sujets sans intervention humaine. Les variantes régionales de roulette et de blackjack complètent une sélection déjà très riche. Les méthodes de paiement instantanées rendent l'expérience particulièrement fluide. Les politiques de confidentialité respectent les standards européens en matière de protection des données. Le mode sombre reposant pour les yeux convient particulièrement aux sessions prolongées. Les tables de blackjack live respectent strictement les règles internationales reconnues. Les jeux se chargent rapidement sur mobile sans nécessiter d'application supplémentaire à installer. Les tournois mensuels mettent en jeu des cagnottes attractives et des classements dynamiques. Le blog de la plateforme propose des articles informatifs sur les stratégies de jeu et les tendances du secteur. Les contacts via réseaux sociaux constituent une alternative pratique pour des questions rapides. Les jackpots progressifs atteignent régulièrement des montants susceptibles de transformer une vie. Les délais de traitement sont communiqués de façon transparente avant chaque opération. Les générateurs de nombres aléatoires sont audités par des organismes indépendants reconnus. La page d'accueil met en évidence les jeux populaires et les promotions du moment. Les variantes de baccarat en direct incluent la version Speed et le Squeeze pour les habitués. Le mode portrait comme le mode paysage sont parfaitement supportés pour le confort visuel. Les promotions hebdomadaires offrent des tours gratuits, des bonus de rechargement et des récompenses surprises. Les niveaux de difficulté ajustables ouvrent l'expérience à tous les profils sans exception. Le souci du détail apparent à chaque écran mérite d'être souligné comme un véritable atout. L'expérience laisse une impression positive durable, signe d'un travail de fond réussi.
Les premières impressions laissées par monsterwin confirment le sérieux du projet et le soin apporté à l'ensemble. Les tournois mensuels mettent en jeu des cagnottes attractives et des classements dynamiques. Les sessions de roulette en direct offrent plusieurs angles de caméra pour une immersion maximale. Les tickets envoyés par e-mail reçoivent une réponse circonstanciée dans des délais courts. Le tableau de bord personnel centralise l'historique de jeu, les bonus et les transactions. L'expérience nomade est aussi complète que la version bureau, sans aucune fonctionnalité retirée. Les options de retrait express permettent aux joueurs pressés de récupérer leurs gains en un temps record. Les mineurs sont strictement écartés grâce à des contrôles d'identité rigoureux. Les statistiques personnelles consultables en ligne aident à mieux comprendre ses habitudes de jeu. Les jeux instantanés et les cartes à gratter complètent agréablement l'offre principale. Les récompenses d'anniversaire ajoutent une touche personnelle appréciée par la communauté. Les variantes de baccarat en direct incluent la version Speed et le Squeeze pour les habitués. Une foire aux questions très complète couvre la majorité des sujets sans intervention humaine. Le moteur de recherche interne retrouve un titre en quelques caractères seulement. Le site s'adapte automatiquement à la taille de l'écran pour conserver lisibilité et confort. Les frais sur les opérations financières sont nuls ou réduits selon le mode choisi. Les politiques de confidentialité respectent les standards européens en matière de protection des données. Cette proposition s'impose progressivement comme une référence solide du jeu francophone. La constance dans la qualité est probablement le meilleur compliment que l'on puisse faire à cette plateforme.
Board of Observers
Cosimo Fiorenza
Vice President & General Counsel
Quantum Valley Investments
Jan Haas
President, BlueIO
W. David Lee
Founder, Lumicell
Mike Ruettgers
Retired Chairman, Lahey Clinic
Former CEO, EMC Corporation
Todd Harrington
Partner, Intuitive Venture Partners
Investors





